WANG Yan, CHEN Yong-mei, WAN Ping-yu*, HAN Yan-ming, QIN Te-fu
(1. School of Science, Beijing University of Chemical Technology, Beijing 100029, China; 2. Research Institute of Wood Industry, Chinese Academy of Forestry, Beijing 100091, China)
Abstract:CaO/MgO composite solid base catalysts were prepared by impregnating calcium acetate on magnesium oxide supports followed by calcination. The surface basicity of the catalysts was dependent on preparation conditions: a surface basicity of up to 30.2 mmol/g was achieved at a CaO/MgO ratio of 0.08 and calcination at 700 °C for 24 h. The as-prepared catalysts were employed to catalyze the cleavage of ether bonds in lignin. Results showed that the weight-average molecular weight (Mw) of lignin degradation products decreased from approximately 3000 to below 800, and the hydroxyl value increased from about 200 mg/g to around 480 mg/g. FT-IR characterization confirmed a significant reduction in ether bonds and a marked increase in hydroxyl groups in the degraded products.
Key words:lignin; solid base catalyst; ether bond
1 Introduction
Lignin, a natural macromolecule second only to cellulose in abundance in the biosphere, is widely distributed in the cell walls of higher plants and represents one of the most important renewable resources. As a major byproduct of the pulp and paper industry, lignin is produced in large quantities. However, its chemical stability and low reactivity render it difficult to utilize without proper treatment. Lignin is typically subjected to graft modification or degradation to enhance its reactivity, enabling its large-scale industrial application in the synthesis of polyurethanes, epoxy resins, phenolic resins, and hydrogels.
Lignin is a macromolecular compound composed of numerous phenylpropane (C9) units linked by C–C bonds (e.g., α–α, β–β) and ether bonds (e.g., α–O–4, β–O–4, β–O–5), among which β-type ether bonds account for more than 50%. Major lignin degradation methods include biological and chemical approaches. Biological fermentation or enzymatic hydrolysis is time-consuming and requires harsh conditions, whereas chemical methods (redox, photocatalysis, electrocatalysis) break chemical bonds in lignin using chemical, light, or electrical energy. The β–O–4 ether bond is one of the weakest linkages between lignin structural units.
Previous studies by Thring, Miller, Karagöz, and others reported the alkaline hydrolysis of ether bonds in lignin. Similar to aqueous alkalis, solid base catalysts can abstract active hydrogen from lignin to initiate ether bond hydrolysis. Compared with homogeneous bases, solid base catalysis offers advantages of high activity, high selectivity, easy product separation, and reusability, with enhanced selectivity for reactions governed by steric or shape-selective effects.
The mechanisms of thermal lignin decomposition catalyzed by solid bases such as calcium carbonate and calcium chloride have been discussed, but these processes require high temperatures and generate few active functional groups. Among solid base catalysts, CaO/MgO composite oxides are promising for industrialization due to abundant raw materials and simple preparation. This work investigates the catalytic hydrolysis of lignin over CaO/MgO composite solid bases and the effects of catalyst properties on lignin degradation.
2 Experimental
2.1 Materials, Reagents and Instruments
Corn stover enzymatic lignin (industrial grade) was supplied by Jilin Chenming Paper Co., Ltd. Phenol, calcium acetate (), magnesium oxide, benzoic acid, anhydrous ethanol, cyclohexane, and acetonitrile were of analytical grade. Deionized water was used throughout.
Equipment: DHG-9053A electric thermostatic drying oven; SX2-2.5-10 muffle furnace; TDL-5A low-speed desktop large-capacity centrifuge; RE-52A rotary evaporator; 848 automatic potentiometric titrator; HR6000 X-ray diffractometer (XRD); ESCALAB 250 X-ray photoelectron spectrometer (XPS); GPC515-2410 gel permeation chromatograph (GPC); Tensor27 Fourier-transform infrared spectrometer (FT-IR).
2.2 Preparation of CaO/MgO Composite Solid Base Catalysts
Exactly 1.0 g of MgO powder pre-calcined at 500 °C was impregnated in 10 mL aqueous calcium acetate solutions of varying concentrations (5.0, 10.0, 15.0, 22.4, 25.0 g/L). After thorough stirring and standing at room temperature for 6 h, the mixture was dried at 80 °C and calcined in a muffle furnace at 500–700 °C for 0.5–24 h. The resulting solid powder was cooled and collected.
Surface basicity was determined by the non-aqueous solvent back-titration method reported in the literature, expressed as mmol of NaOH equivalent per gram of solid base catalyst. The powders were characterized by XRD and XPS.
2.3 Solid Base-Catalyzed Lignin Degradation
A 4.0 g lignin sample was dissolved in 160 mL ethanol–water (1:1 v/v) with stirring. Then 0.4 g CaO/MgO catalyst and 0.2 g phenol (as a polymerization inhibitor) were added. The mixture was transferred to a 100 mL autoclave and heated at 170 °C for 4.5 h. After cooling to room temperature, the reaction mixture was concentrated to ~5 mL by rotary evaporation, dried at 80 °C, and analyzed for molecular weight, hydroxyl value, and FT-IR.
Hydroxyl values (VOH) of lignin and its products were measured according to ASTM-E1899-2008 for polyether polyols, expressed as mg KOH equivalent per gram of sample.
3 Results and Discussion
3.1 Mechanism of Lignin Ether Bond Hydrolysis over Solid Base Catalysts
Thring et al. proposed the mechanism of alkaline lignin hydrolysis: phenolic hydroxyl groups in lignin are deprotonated to phenoxide anions under alkaline conditions, triggering a series of electron transfers that ultimately cleave ether bonds to form small fragments. Basic sites on the solid base catalyst surface abstract phenolic hydroxyl hydrogen from lignin, initiating the same electron-transfer pathway and leading to ether bond cleavage and lignin degradation. The number of active sites (quantified as surface basicity) strongly influences the hydrolysis of lignin ether bonds.
3.2 Factors Affecting Surface Basicity of CaO/MgO Composite Catalysts
CaO/MgO composites were obtained by high-temperature calcination of -impregnated MgO. XRD patterns of –MgO powders calcined at different temperatures show only MgO diffraction peaks at 500 °C, indicating calcium acetate decomposed into amorphous products. diffraction peaks appeared at 600 °C, and CaO peaks (2θ ≈ 37°, 54°) emerged at 700 °C, confirming sequential decomposition: calcium acetate → calcium carbonate → calcium oxide.
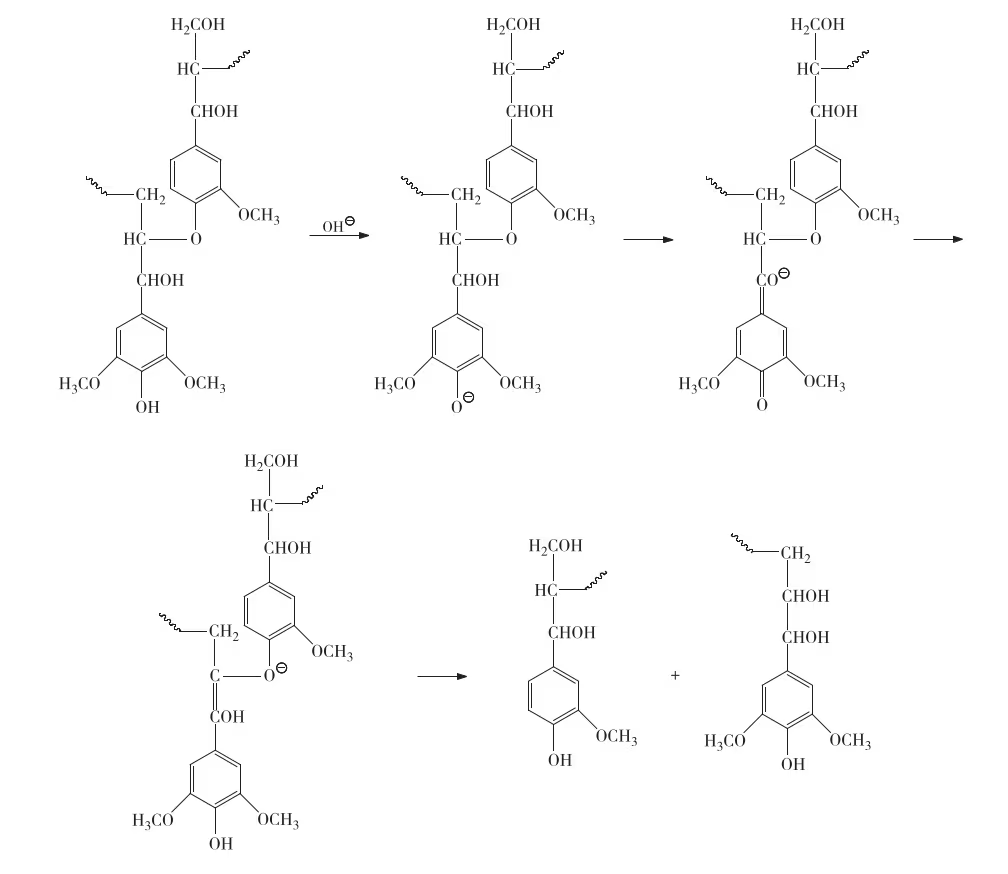
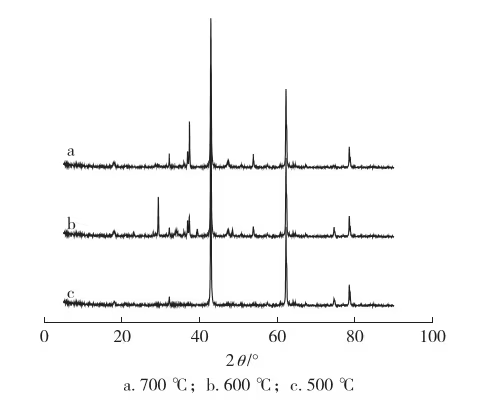
A diffraction peak at 2θ ≈ 32°, assigned to a mixed crystal phase formed by and doping into MgO or CaO lattices, intensified with increasing calcination time.
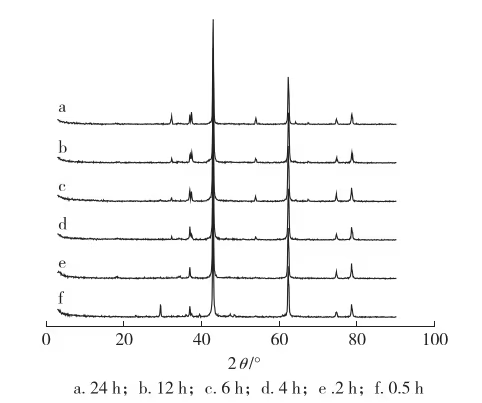
XPS O1s spectra of the calcined products show two peaks: one at 531.8 eV assigned to and another at 529.6 eV assigned to . Both binding energies are slightly higher than those reported for pure MgO and CaO, verifying the formation of a CaO/MgO mixed crystal phase.
Surface basicity increased with CaO/MgO mass ratio, calcination temperature, and calcination time. Formation of the surface mixed crystal promotes the generation of basic active sites (exposed lattice oxide ions ), increasing surface basicity. The highest basicity (30.2 mmol/g) was achieved at CaO/MgO = 0.08, 700 °C, 24 h.
Table 1 Effects of preparation conditions on surface basicity of catalysts
| No. | CaO/MgO | Calcination temperature / °C | Calcination time / h | Surface basicity / (mmol·g⁻¹) |
|---|---|---|---|---|
| 1 | 0.02 | 700 | 24 | 21.8 |
| 2 | 0.05 | 700 | 24 | 24.9 |
| 3 | 0.07 | 700 | 24 | 28.6 |
| 4 | 0.08 | 700 | 24 | 30.2 |
| 5 | 0.09 | 700 | 24 | 29.7 |
| 6 | 0.08 | 300 | 24 | 19.2 |
| 7 | 0.08 | 500 | 24 | 25.3 |
| 8 | 0.08 | 600 | 24 | 27.1 |
| 9 | 0.08 | 700 | 2 | 16.7 |
| 10 | 0.08 | 700 | 4 | 17.8 |
| 11 | 0.08 | 700 | 6 | 20.1 |
| 12 | 0.08 | 700 | 12 | 23.0 |
| 13 | 0.08 | 700 | 18 | 25.1 |
| 14 | 0.08 | 700 | 26 | 28.4 |
3.3 Lignin Degradation Catalyzed by CaO/MgO Composite
3.3.1 Molecular Weight and Hydroxyl Value of Degraded Products
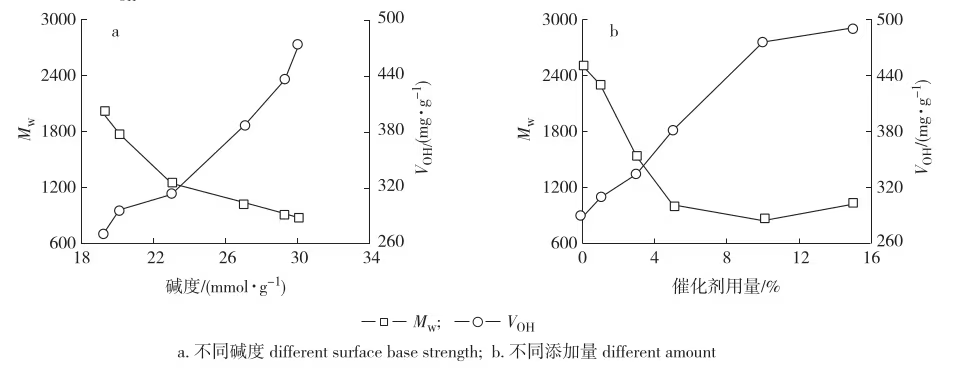
Raw lignin had a weight-average molecular weight (Mw) of ~3000 and hydroxyl value (VOH) of ~200 mg/g. Alkaline cleavage of ether bonds reduces molecular weight and increases hydroxyl value. Lignin degraded to Mw < 1000 and VOH > 450 mg/g can replace polyether polyols in polyurethane synthesis.
After reaction in ethanol–water at 170 °C with CaO/MgO, Mw decreased sharply and VOH increased markedly. At the same catalyst dosage, higher basicity yielded lower Mw and higher VOH, indicating better degradation. At a dosage of 10% (based on lignin), the catalyst with 30.2 mmol/g basicity reduced Mw to ~800 (polydispersity index from 3.7 to 2.4) and increased VOH to ~480 mg/g. Degradation improved with catalyst loading but leveled off above 10%, consistent with the dependence on surface basic sites: more sites promote ether bond cleavage, lowering Mw and raising VOH.
3.3.2 Structural Characterization of Lignin Degradation Products
SEM images show that raw corn stover enzymatic lignin had a dense surface, whereas degraded particles were significantly smaller with numerous open pores.




Fig. 6 SEM images of lignin sample before and after degradation
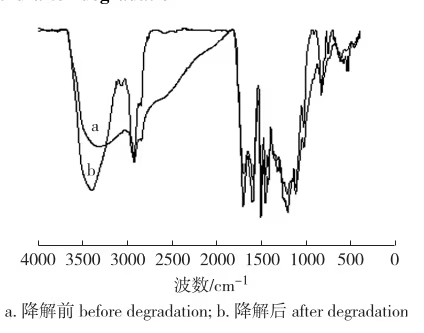
FT-IR spectra of lignin before and after catalysis show:
- 3408 cm⁻¹: –OH stretching
- 1710 cm⁻¹: C=O stretching
- 1460–1610 cm⁻¹: benzene ring characteristic peaks
- 1120 cm⁻¹: ether bond absorption
Benzene rings remain stable during alkaline hydrolysis; relative changes in functional groups were evaluated against the 1510 cm⁻¹ peak. Degraded products showed increased hydroxyl and carbonyl groups and decreased ether bonds, confirming hydrolysis of ether bonds to form hydroxyls, consistent with lower Mw and higher VOH.
4 Conclusions
- CaO/MgO composite solid base catalysts were prepared by impregnating on MgO supports followed by high-temperature calcination. Surface basicity was controlled by CaO/MgO ratio, calcination temperature, and time. A maximum basicity of 30.2 mmol/g was obtained at CaO/MgO = 0.08, 700 °C, 24 h.
- Under optimized catalysis, raw lignin Mw decreased from ~3000 to < 800 and VOH rose from ~200 to ~480 mg/g. FT-IR and SEM confirmed reduced ether bonds, increased hydroxyls, smaller particle size, porous morphology, and reduced polymerization. The degraded lignin can substitute polyether polyols in polyurethane synthesis.
