Keywords: Magnesium Oxide, Oxygen Vacancies, Insulator, Stomach Medicine, Quantum Tunneling
In daily life, Magnesium Oxide (MgO) serves as an antacid and a mild laxative, relieving gastric discomfort and promoting intestinal health; it is also an essential nutritional supplement for the human body. In material science, the rock-salt structure of MgO endows it with excellent mechanical strength, insulation, and thermal conductivity, making it an indispensable material in nanotechnology. It is widely used in electronic components as insulating layers, heat dissipation layers, and in Magnetic Tunnel Junctions (MTJ). Recent research in 2024 has revealed that by regulating oxygen vacancies on the MgO surface, its catalytic activity in fuel cells can be enhanced, accelerating the Oxygen Reduction Reaction (ORR) and increasing fuel cell conversion efficiency, thereby advancing green energy technology. However, with the progress of nanotechnology, controlling defects in MgO at microscopic scales remains a significant challenge.
Have you ever wondered if the common antacid in your medicine cabinet could be related to cutting-edge electronic technology? A substance called Magnesium Oxide (MgO) plays a role in everything from relieving stomach acid to nanotechnology. What kind of “eighteen martial arts” skills does MgO possess that allow it to be so widely applied across such diverse fields?
MgO as a Medical Treatment
When MgO is mentioned, many people immediately think of medical supplies, especially drugs related to the digestive system. MgO is a weakly alkaline substance capable of neutralizing acids. It reacts with hydrochloric acid (HCl) to produce MgCl2 and water (H2O), which is why it is frequently used to neutralize stomach acid and alleviate symptoms of gastroesophageal reflux. Furthermore, in the intestines, MgO dissolves into Mg2+ and OH− states, increasing osmotic pressure. This draws water into the intestinal tract, causing the contents—the stool—to bind with more water, thereby achieving a stool-softening effect. Consequently, MgO is also used as a mild laxative to aid smooth bowel movements. Additionally, MgO is common in various nutritional supplements. Magnesium is an essential mineral for the human body, participating in numerous enzymatic reactions and significantly contributing to nerve function, muscle contraction, and bone health. For those who exercise frequently, moderate MgO supplementation can help relieve muscle cramps and improve athletic performance.

(Figure 1: Illustration of stomach medicine | Source: Wikimedia Commons)
Applications of MgO in Material Science
MgO possesses a cubic rock-salt structure with a lattice constant of approximately 4.21 Å. Each magnesium ion (Mg2+) is coordinated with six oxygen ions (O2−), forming a stable three-dimensional crystal. This highly symmetrical and tightly packed structure gives MgO excellent mechanical strength, allowing it to resist deformation from external forces. Therefore, MgO maintains stability under high temperature and high pressure without easily decomposing, leading to its application in fireproof materials and high-temperature insulating layers.
In terms of electrical properties, MgO is a wide-bandgap material with a bandgap of approximately 7.8 eV, classifying it as an insulator. Even in high-temperature and high-electric-field environments, MgO maintains excellent insulation. This makes it widely used in electronic components; for instance, in Field-Effect Transistors (FET), MgO acts as an insulating layer between circuit elements to prevent current leakage, enhancing component stability and efficiency. Moreover, because MgO can withstand high electric fields without conducting, it ensures that circuits operate stably under high-voltage conditions, improving performance and lifespan.
Beyond traditional electronic components, MgO serves as a tunneling insulating layer in Magnetic Tunnel Junctions (MTJ) for spintronic devices. MTJs utilize the quantum tunneling effect (an effect where particles have a probability of passing through an insulating barrier). Because MgO has an ordered lattice structure, it improves matching with device electrodes, significantly increasing tunneling efficiency. This makes MgO a key material for next-generation non-volatile memory, such as MRAM.

Interestingly, while MgO does not conduct electricity, it is an excellent thermal conductor. MgO has a high thermal conductivity—approximately 60 W/mK at room temperature—and high thermal stability. While acting as an insulating layer, it simultaneously serves as a heat dissipation layer, helping components avoid performance degradation caused by high temperatures. Thus, MgO is frequently applied in high-power semiconductors and LEDs.
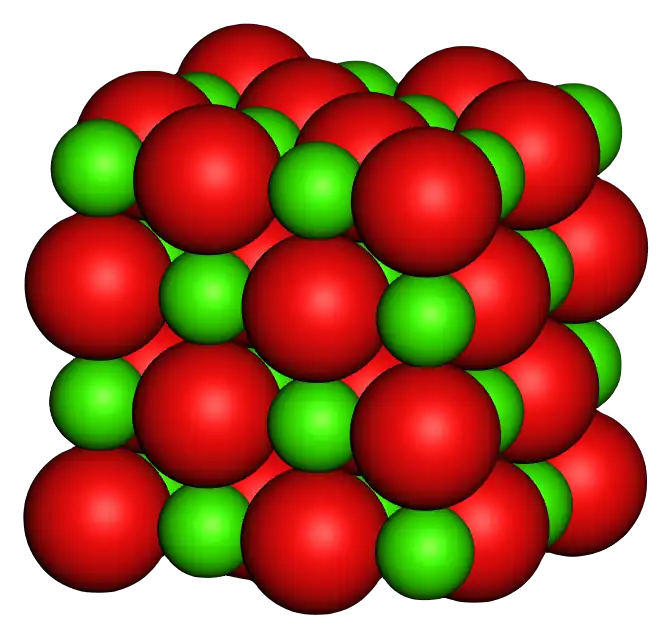
(Figure 2: Schematic of the cubic rock-salt structure of MgO | Source: Wikimedia Commons)
The Latest Research on MgO
MgO continues to be a subject of intense scientific study. A 2024 study in the Journal of Applied Physics pointed out that different thermal treatments can regulate the generation of oxygen vacancies in MgO. Oxygen vacancies refer to missing oxygen atoms in the crystal lattice where they should be present. These vacancies change the bandgap of MgO and affect its insulating properties. Furthermore, appropriate oxygen vacancies can enhance the polarization capability of MgO, thereby increasing its capacitance and allowing it to store more charge. It appears that “defective” MgO is not necessarily inferior to “perfect” MgO! Additionally, a 2024 study in Science Advances indicated that surface defects on MgO crystals can effectively help oxygen molecules dissociate into oxygen atoms. These defects lower the activation energy of the Oxygen Reduction Reaction (ORR), thereby increasing the reaction rate. This discovery promotes the development of fuel cells, where oxygen molecules combine with protons (H+) and electrons (e−) at the cathode to generate water (H2O
). This process typically has high activation energy, but by regulating surface defects on MgO, the decomposition of oxygen molecules into O2− can be promoted, accelerating the entire reaction and improving fuel cell efficiency.
MgO has been widely utilized in both medical pharmaceuticals and the electronics industry. However, as nanotechnology strives for thinner and smaller components, MgO faces certain limitations. As the volume of MgO shrinks, material defects increase significantly. Although defects can lead to unexpected physical properties, they also become uncontrollable variables. Systematically controlling the ratio of defects remains a major challenge in fabrication. Nevertheless, this environmentally and human-friendly substance is undoubtedly a material worthy of continued in-depth research by scientists!
