Synthetic materials are currently widely used in transportation, transportation, construction, electronics, electronics, chemical industry and other fields, and play a huge role in the construction of national economic construction. However, synthetic materials are generally flammable. In order to solve and eliminate this hidden danger, flame retardants came into being. In addition to the flame retardant effect in the synthetic material, in the synthetic material, the effect of inhibiting smoking and hydrogen chloride production is also given non -toxic and non -corrosive, which is widely used.
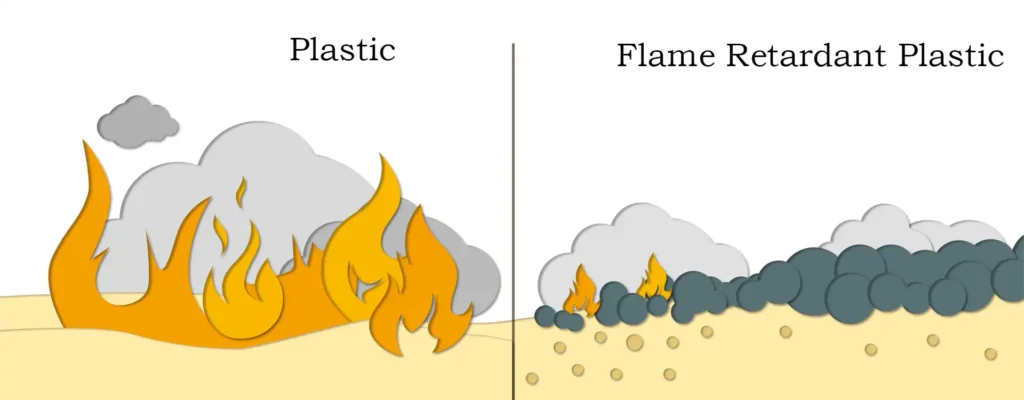
Today, where environmental protection requirements are increasingly strengthened, inorganic flame retardants show strong competitiveness and development potential. Among them, magnesium hydroxide is an excellent variety of inorganic flame retardants. Magnesium hydroxide is a added inorganic flame retardant. Compared with similar inorganic flame retardants, it has a better smoke suppression effect. Because 80 % of the fire suffocated by smoke, “smoke suppression” in contemporary flame retardant technology is more important than “flame retardant”.
Magnesium hydroxide is discharged from harmful substances during production, use, and abandonment, and it can also neutralize the acidic and corrosive gases produced during the combustion process. It is an environmentally friendly green flame retardant. The heat decomposition temperature of magnesium hydroxide hydroxide is high, which is 140 ° C higher than the current commonly used inorganic flame retardant hydroxide, which can enable the synthetic material with magnesium hydroxide to withstand higher processing temperature, which is conducive to accelerating the squeezing speed and shortening the mold molding. Time also helps improve flame retardant efficiency. The particle size of magnesium hydroxide is small, and the equipment is small, which is conducive to extending the service life of the processing equipment. At the same time, the raw materials are easy to obtain and the production cost is low. Fields.
Due to the many excellent characteristics of magnesium hydroxide, the basic research and application research on magnesium hydroxide at home and abroad within the past 10 years is very active. At present, more than 20 companies in nearly 10 countries have produced more than 20 varieties. About 170,000 tons. Many countries are still being constructed or planned to build new magnesium hydroxide devices. According to relevant data reports, the total annual capacity capacity of magnesium hydroxide device will be constructed in recent years is about 140,000 tons. At present, my country’s annual production capacity of magnesium hydroxide flame retardant is about 13,000 tons. The main manufacturers include Shandong Jiaozhou Guhe Magnesium Salt Factory, Shanghai Zhentai Chemical Factory, Wuyi County Flame Reserve Factory, Jiangsu Nanhua Group , Shandong Haihua Group, Wenzhou Potassium Fertilizer Factory, etc. Compared with the advanced level of foreign countries, my country’s enterprises are small, small, and low in technology, and urgently need to improve the overall level of the industry.
At present, the main industrialized synthesis methods of magnesium hydroxide include magnesium chloride thermal solution and brine ammonia. There are two major problems in the production process of magnesium hydroxide. One is that it is easier to manufacture ordinary magnesium hydroxide. The sedimentary particles are thinner and large, and it is difficult to process after washing and separation. Therefore, the current problem that is urgent to solve is the study of the impact of the pineral and precipitation on product performance during the production process. Through modified research, products with different physical indicators are produced.
Improve the existing preparation process, prepare needle -shaped or shear -shaped magnesium hydroxide, and improve the distortion strength and extension rate of materials. For example, the continuous settlement method of brine ammonia is improved as follows: refined brine and excess ammonia mixed, react under high temperature and high pressure, prepare magnesium hydroxide floating fluid, and then separate and washed. , Surface treatment, filtering, and drying to obtain magnesium hydroxide hydroxide, control the reaction conditions, can obtain products of different specifications; for the surface modification research of magnesium hydroxide, choose excellent surface modifiers with excellent performance, so that the surface of the particles can make the surface of the particles surface. Improve activity, improve dispersability, improve flame retardant effects, compatibility with polymer materials, resistance and thermal performance, and to expand production scale, improve economic benefits, and expand the field of application.
Due to the enhanced awareness of environmental protection, inorganic flame retardants have been used abroad. Among them, the consumption of inorganic flame retardant consumption in the United States, Japan, and Western Europe accounts for 60 %, 64 %, and 50 % of the total consumption of the total flame retardant, respectively. The inorganic flame retardant is mainly aluminum hydroxide and magnesium hydroxide. Because magnesium hydroxide has many advantages compared with aluminum dioxide, magnesium hydroxide accounted for increasingly and larger. According to foreign data statistics, the consumption consumption of magnesium hydroxide flame retardant in Western developed countries accounts for more than 30 % of the consumption of inorganic flame retardants. The current annual consumption of the United States, Western Europe, and Japan is about 50,000 tons, 80,000 tons, and 30,000 tons, respectively. In the next 5 years, the average annual growth rate of the United States, Western Europe, and Japan will be 12 %, 8 %, and 7 %, respectively. Based on the above analysis, the development of the world’s magnesium hydroxide flame retardant is bright.
In recent years, my country’s synthetic polymer materials have developed extremely rapidly. It is expected that plastic production will exceed 8 million tons in 2002. According to the experience of developed countries, the annual amount of plastic flame retardant will reach more than 600,000 tons. If the inorganic flame retardant accounts for 50 %, and the magnesium hydroxide flame retardant accounts for about 30 % of the inorganic flame retardant, hydroxide is required every year The magnesium flame retardant is 90,000 tons, which shows that my country’s magnesium hydroxide development potential is huge.
The world’s environmental protection requirements are getting higher and higher, and magnesium hydroxide of inorganic flame retardants who are friendly to environmental friendly are valued by countries. Relevant research, cooperative development, and production activities are very active. Developed countries have invested heavily in research and development. In addition to general varieties, various special and complex new products have emerged endlessly, and the application fields have been continuously developed. Magnesium hydroxide flame retardant has been widely used in various plastic products, especially high -performance magnesium hydroxide flame retardants for electric wires and cables. It is welcomed by users.
With the rapid development of my country’s synthetic polymer material industry and the continuous improvement of flame retardant regulations, the demand for flame retardant has increased, and the demand for magnesium hydroxide hydroxide hydroxide hydroxide hydroxide hydroxide in non -toxic and smoke is very urgent. my country is also a large country of magnesium ore resources, with unique resource advantages and good market prospects. Therefore, my country should improve the existing production technology, large -scale production, and strengthen the research on the application of magnesium hydroxide, promote the production and development of my country’s magnesium hydroxide flame retardant, meet the needs of the rapid development of the plastic industry in my country, while accelerating the flame retardant in my country Adjustment of industrial product structure.

