Magnesium Hydroxide: Properties, Uses, and Market Outlook
Properties of Magnesium Hydroxide
Magnesium hydroxide (Mg(OH)₂) is an inorganic compound commonly found as a white powder or crystalline solid. It has a molecular weight of 58.32 g/mol, a density of 2.36 g/cm³, and a melting point of approximately 350°C. At high temperatures, it decomposes into magnesium oxide and water. Magnesium hydroxide has low water solubility (0.009 g/100 mL water at 20°C), making it sparingly soluble. It exhibits high thermal stability and excellent flame retardant properties. Its chemical properties are relatively stable at room temperature, showing little reactivity with acids or bases. However, at high temperatures, it reacts with acids, bases, and salts to form corresponding compounds.
Uses of Magnesium Hydroxide
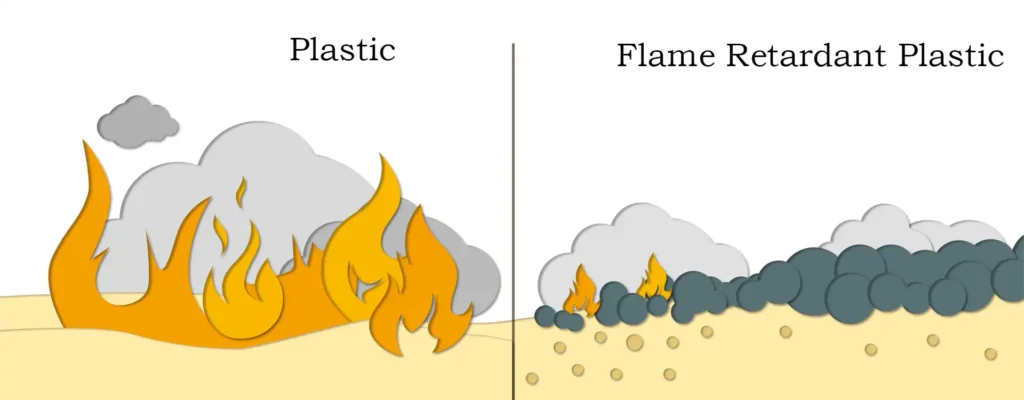
Magnesium hydroxide has wide applications in industry and daily life. It’s a crucial flame retardant and fireproofing material, effectively suppressing flame propagation and reducing smoke generation by releasing water vapor at high temperatures. It’s widely used in environmental protection as a flue gas desulfurizer and wastewater treatment agent, neutralizing acidic wastewater and removing heavy metal ions. In medicine, it serves as an antacid and laxative, treating hyperacidity and constipation. It also finds applications in rubber, plastics, papermaking, and ceramics, enhancing material properties.
Upstream Raw Materials of Magnesium Hydroxide
The production of magnesium hydroxide primarily relies on two upstream raw materials: magnesium ore and water. Magnesium ores, such as dolomite and magnesite, are the main sources. Calcination of these ores yields magnesium oxide, which then reacts with water to produce magnesium hydroxide. Seawater is also a significant magnesium source; magnesium hydroxide can be extracted through seawater desalination and chemical precipitation. The purity and quantity of water directly impact product quality and production efficiency, making water source selection and management crucial.
Downstream Products of Magnesium Hydroxide
Magnesium hydroxide has diverse downstream products, primarily used in various industrial sectors. In the rubber and plastics industry, it acts as a filler and reinforcing agent, improving strength, heat resistance, and flame retardancy. In pharmaceuticals and health products, it’s a common active ingredient, particularly in antacid and laxative formulations. In building materials, it shows promise as an environmentally friendly flame retardant and fireproofing agent. The development and application of these downstream products continuously drive market demand and industry growth.
Production Processes of Magnesium Hydroxide
Two main methods produce magnesium hydroxide: wet and dry processes. The wet process involves chemical reactions, such as suspending magnesium oxide in water to form a magnesium hydroxide slurry, followed by precipitation, filtration, and drying. The dry process involves high-temperature calcination of magnesite or dolomite to obtain magnesium oxide, which then reacts with water under high temperature and pressure to produce magnesium hydroxide. The wet process offers lower costs and suits mass production, while the dry process yields higher purity products, suitable for applications demanding high quality.
Storage Methods of Magnesium Hydroxide
Storing magnesium hydroxide requires specific environmental conditions to maintain quality. It should be stored in a dry, well-ventilated warehouse, protected from moisture, as it readily absorbs moisture and cakes, affecting its physical properties and performance. The storage environment should avoid high temperatures and direct sunlight to prevent decomposition and performance degradation. Airtight containers, such as plastic bags or drums, should be used to minimize air contact. Avoid storing it with acidic substances or strong oxidizing agents to prevent unwanted chemical reactions.
Current Market Status and Future Outlook of Magnesium Hydroxide
The magnesium hydroxide market has shown steady growth in recent years, driven by environmental regulations and increasing demand for flame retardant materials. Stringent global environmental regulations are expanding its use in plastics, rubber, and building materials as an eco-friendly flame retardant. Its applications in flue gas desulfurization and wastewater treatment are also growing, further increasing market demand. Future technological advancements and process improvements are expected to lower production costs and enhance product performance, promising a bright market outlook.
Conclusion
Magnesium hydroxide, a vital inorganic compound, plays a significant role across various fields due to its unique physicochemical properties. Analyzing its properties, uses, upstream and downstream materials, production processes, and storage methods reveals its importance in industrial production and its broad applications in environmental protection and medicine. Continued technological progress and expanding market demand will create further growth opportunities for the magnesium hydroxide industry.
